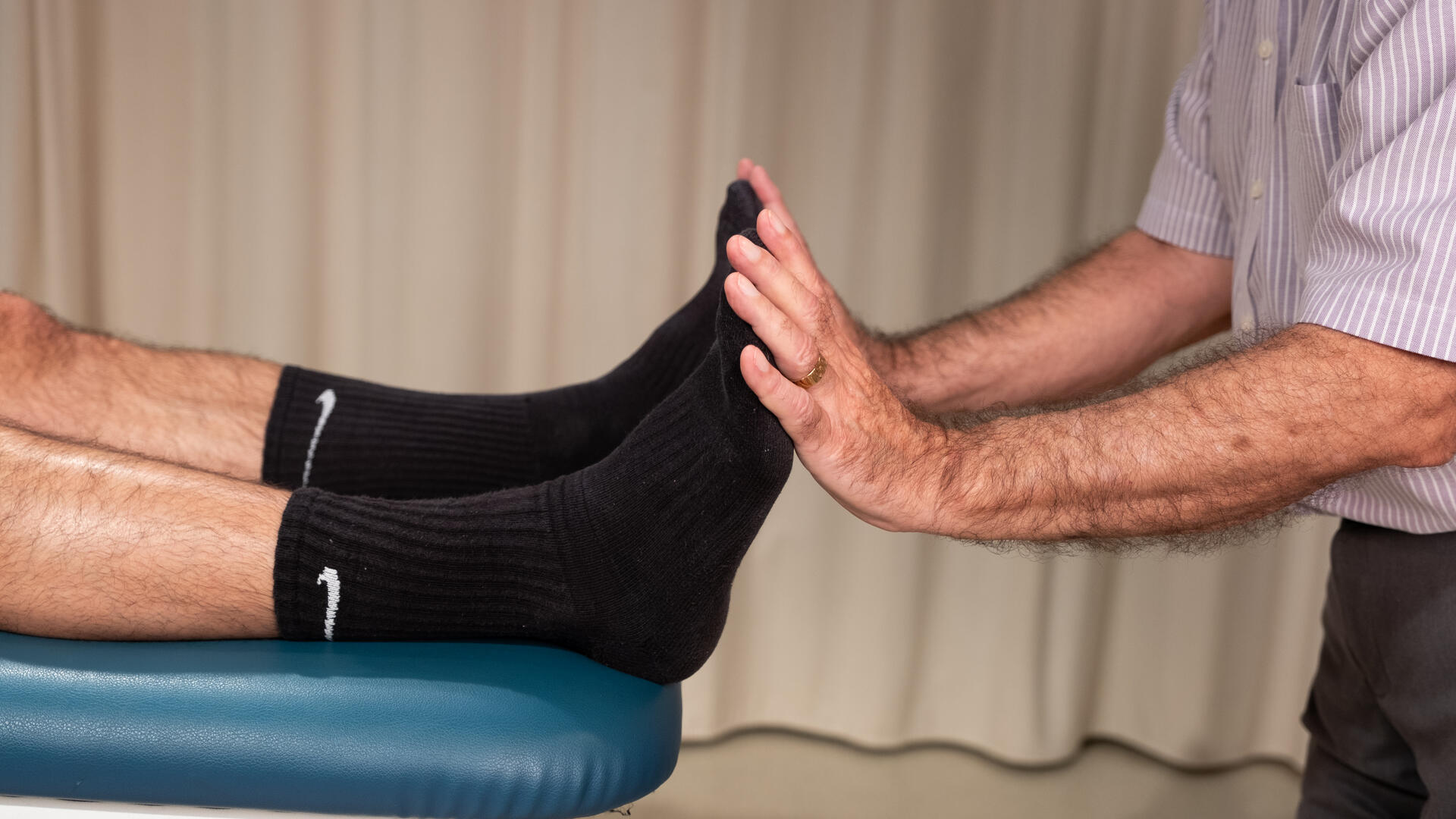
Trial Purpose
The purpose of this study is to learn if lymphatic osteopathic manipulative treatment (OMT) is effective in stimulating a stronger immune system than exercise or light touch sham treatment alone in healthy people. This study will investigate all three groups and help us to identify which immune cells and cytokines are most affected by each experimental intervention.
View full trial information on ClincalTrials.gov
National Trial Reference Number
NCT06565065

Eligibility
Our certified osteopathic physicians will assess if you qualify to take part in this clinical trial, but this information can be used to see if you may be eligible and should schedule an appointment to meet one of our doctors.
- If you meet these requirements and are interested, please use the following link to pre-screen:
Pre-Screen Now
Gender |
Healthy Volunteers |
Age Range |
|---|---|---|
| All | Yes | 18+ |
Inclusion Criteria:
- Individuals must be 18 years or older.
- Individuals must provide informed consent.
- Individuals must be healthy with a BMI under 30, with no primary or secondary immune deficiency or autoimmune disease/must not be immunocompromised.
- Individuals must not be using or recently have used any immune-altering medications such as corticosteroids, cyclosporin, tacrolimus, and pimecrolimus
- Individuals must not have had lymphatic OMT or other physical manipulation such as acupuncture, physical therapy, chiropractic, or massage therapy in the previous four weeks before study enrollment.
- Individuals must have the ability to receive OMT with lymphatic techniques.
- Individuals must be able to tolerate 12 minutes of moderate physical activity.
- Individuals must not be pregnant or breastfeeding.
- Individuals must not have had a splenectomy or other spleen or oncologic surgery within the past 6 months.
- Individuals must be infection-free for the past 6 weeks before starting the study and have not received any immunizations within the past 6 weeks.
- Individuals must be willing to avoid strenuous activity at least 24 hours before each visit.
- Individuals must be willing to abstain from moderate to intense activity in the 4 hours between their 2nd blood draw and their final blood draw at each of their three visits.
Exclusion Criteria:
- Individuals are younger than 18 years of age.
- Individuals who do not provide informed consent.
- Individuals with a BMI over 30, or with primary or secondary immune deficiency or autoimmune disease or is immunocompromised.
- Individuals using or recently have used any immune-altering medications such as corticosteroids, cyclosporin, tacrolimus, and pimecrolimus
- Individuals who have had lymphatic OMT or other physical manipulation such as acupuncture, physical therapy, chiropractic, or massage therapy in the previous four weeks before study enrollment.
- Individuals who don't have the ability to receive OMT with lymphatic techniques.
- Individuals who cannot tolerate 12 minutes of moderate physical activity.
- Individuals who are pregnant or breastfeeding.
- Individuals who have had a splenectomy or other spleen or oncologic surgery within the past 6 months.
- Individuals who have had an infection in the 6 weeks before starting the study and or have received any immunizations within the past 6 weeks.
- Individuals not willing to avoid strenuous activity at least 24 hours before each visit.
- Individuals not willing to abstain from moderate to intense activity in the 4 hours between their 2nd blood draw and their final blood draw at each of their three visits.
IRB Protocol No.
P24/IRB/002
Category
Osteopathic Manipulative Medicine (OMM)
Immunology
Principal Investigator

Brian Loveless, DO
NMM/OMM and Family Medicine
Location
Western University of Health Sciences
Patient Care Center
For detailed eligibility,
please visit: ClinicalTrials.gov